Development and validation of a robust QbD-guided (RP-HPLC) analytical technique for quantifying coenzyme Q10 in pharmaceutical dosage form
DOI:
https://doi.org/10.69857/joapr.v13i5.1696Keywords:
Coenzyme Q10, RP-HPLC, Quality by Design (CCD), Method validationAbstract
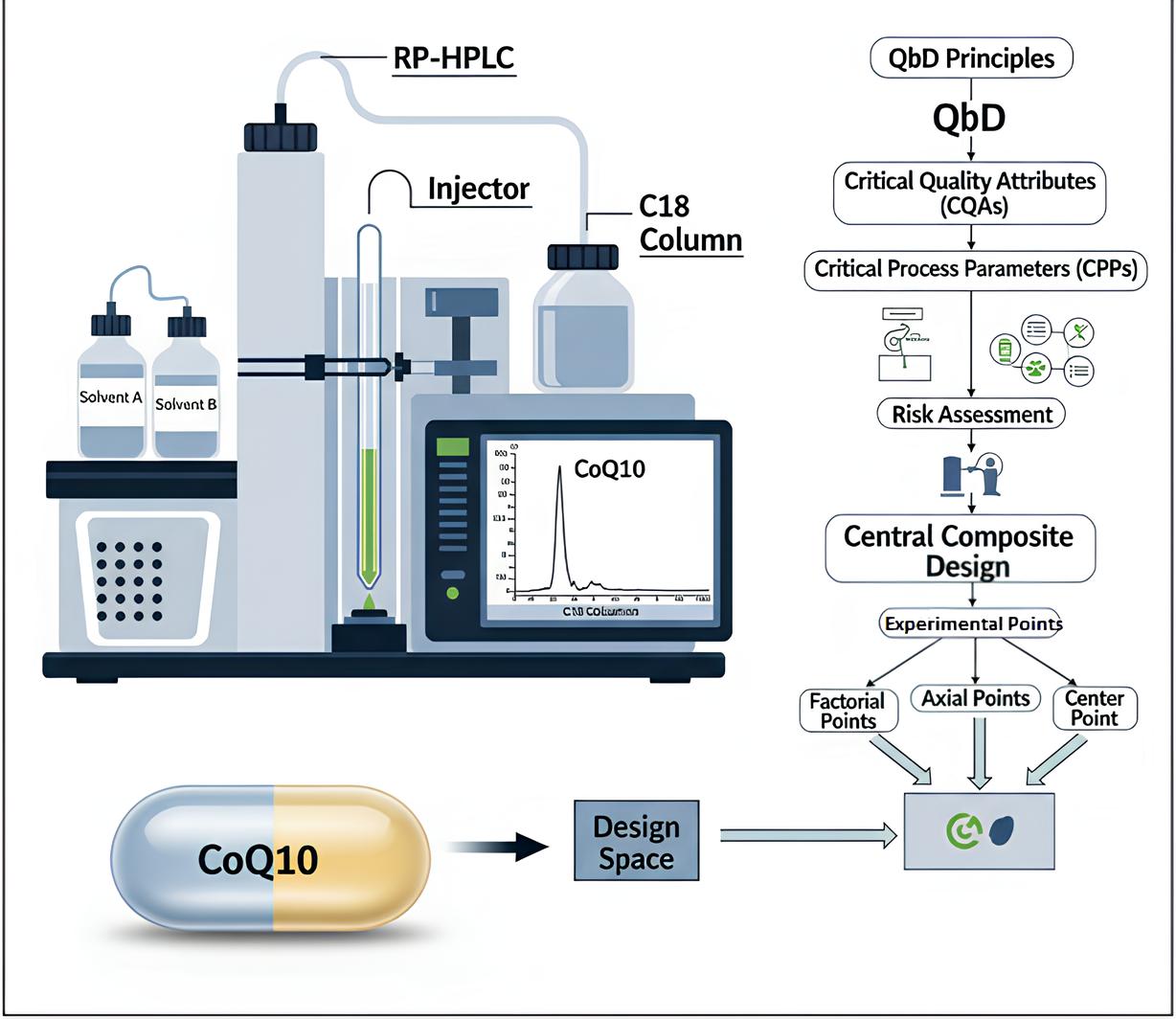
Background: Coenzyme Q10 (CoQ10) is a lipophilic antioxidant that contributes to mitochondrial energy production but poses analytical challenges due to low solubility and oxidative sensitivity. An accurate and validated method is essential to ensure quality control of its pharmaceutical dosage forms. Methodology: A reverse-phase high-performance liquid chromatography (RP-HPLC) method was designed using Quality by Design (QbD) principles. A Central Composite Design was employed to study the influence of critical parameters, including flow rate, gradient time, and mobile phase ratio. Separation was carried out on a Gemini C18 column (250 × 4.6 mm, 5 µm) with gradient elution using ethyl acetate: acetonitrile (50:50) and methanol: acetonitrile (80:20) containing 0.1% ammonia. The method was validated in accordance with ICH-Q2 (R1) guidelines. Results: The optimized conditions yielded a sharp CoQ10 peak at approximately 12.9 minutes with high resolution (Rs > 36), theoretical plates (~33,800), and acceptable tailing (≤1.3). Linearity was established over 2.5–200 µg/mL (r² = 0.9997). Accuracy ranged from 99.2–101.4%, precision was within %RSD ≤1.5%, and robustness was demonstrated under deliberate variations. Discussion: The method proved capable of consistently quantifying CoQ10 with superior specificity and sensitivity. Application to commercial soft gel formulations confirmed assay values at 99.5% of the label claim, meeting pharmacopeial standards. Conclusion: This QbD-based RP-HPLC method offers a validated, reproducible, and regulation-compliant strategy for the quality control and stability assessment of CoQ10 dosage forms, with potential extension to similar pharmaceutical compounds.
Downloads
References
Kowalczyk P, Sulejczak D, Kleczkowska P, Bukowska-Ośko I, Kucia M, Popiel M, Wietrak E, Kramkowski K, Wrzosek K, Kaczyńska K. Mitochondrial Oxidative Stress—A Causative Factor and Therapeutic Target in Many Diseases. Int J Mol Sci, 22, 13384 (2021) https://doi.org/10.3390/ijms222413384.
Vinarov Z, Abdallah M, Agundez JAG, Allegaert K, Basit AW, Braeckmans M, Ceulemans J, Corsetti M, Griffin BT, Grimm M, Keszthelyi D, Koziolek M, Madla CM, Matthys C, McCoubrey LE, Mitra A, Reppas C, Stappaerts J, Steenackers N, Trevaskis NL, Vanuytsel T, Vertzoni M, Weitschies W, Wilson C, Augustijns P. Impact of gastrointestinal tract variability on oral drug absorption and pharmacokinetics: An UNGAP review. European Journal of Pharmaceutical Sciences, 162, 105812 (2021) https://doi.org/10.1016/j.ejps.2021.105812.
Prajapati PB, Jayswal K V, Shah SA. DoE and Risk-Based DMAIC Principle for Implementation of Enhanced Analytical Quality by Design Approach to Multipurpose-Chromatography Method for Simultaneous Estimation of Multiple Fixed-Dose Combination Products of Aspirin. J AOAC Int, 104, 1430–41 (2021) https://doi.org/10.1093/jaoacint/qsab058.
ter Horst JP, Turimella SL, Metsers F, Zwiers A. Implementation of Quality by Design (QbD) Principles in Regulatory Dossiers of Medicinal Products in the European Union (EU) Between 2014 and 2019. Ther Innov Regul Sci, 55, 583–90 (2021) https://doi.org/10.1007/s43441-020-00254-9.
Marcheggiani F, Cirilli I, Orlando P, Silvestri S, Vogelsang A, Knott A, Blatt T, Weise JM, Tiano L. Modulation of Coenzyme Q10 content and oxidative status in human dermal fibroblasts using HMG-CoA reductase inhibitor over a broad range of concentrations. From mitohormesis to mitochondrial dysfunction and accelerated aging. Aging, 11, 2565–82 (2019) https://doi.org/10.18632/aging.101926.
Saini S, Sharma T, Patel A, Kaur R, Tripathi SK, Katare OP, Singh B. QbD-steered development and validation of an RP-HPLC method for quantification of ferulic acid: Rational application of chemometric tools. Journal of Chromatography B, 1155, 122300 (2020) https://doi.org/10.1016/j.jchromb.2020.122300.
Subriadi AP, Najwa NF. The consistency analysis of failure mode and effect analysis (FMEA) in information technology risk assessment. Heliyon, 6, e03161 (2020) https://doi.org/10.1016/j.heliyon.2020.e03161.
Bhatti GK, Gupta A, Pahwa P, Khullar N, Singh S, Navik U, Kumar S, Mastana SS, Reddy AP, Reddy PH, Bhatti JS. Targeting mitochondrial bioenergetics as a promising therapeutic strategy in metabolic and neurodegenerative diseases. Biomed J, 45, 733–48 (2022) https://doi.org/10.1016/j.bj.2022.05.002.
Verch T, Campa C, Chéry CC, Frenkel R, Graul T, Jaya N, Nakhle B, Springall J, Starkey J, Wypych J, Ranheim T. Analytical Quality by Design, Life Cycle Management, and Method Control. AAPS J, 24, 34 (2022) https://doi.org/10.1208/s12248-022-00685-2.
Dewi M, Pratama R, Arifka M, Chaerunisaa A. Quality By Design: Approach to Analytical Method Validation. Sciences of Pharmacy, 1, 38–46 (2022) https://doi.org/10.58920/sciphar01010033.
Sha’at M, Spac AF, Stoleriu I, Bujor A, Cretan MS, Hartan M, Ochiuz L. Implementation of QbD Approach to the Analytical Method Development and Validation for the Estimation of Metformin Hydrochloride in Tablet Dosage Forms by HPLC. Pharmaceutics, 14, 1187 (2022) https://doi.org/10.3390/pharmaceutics14061187.
Yadav K, Jatain I, Dubey KK, Nitharwal RG, Kaur I. Development of a thin layer chromatographic method for the determination of coenzyme Q 10 produced by Agrobacterium tumefaciens. Sep Sci Plus, 6, (2023) https://doi.org/10.1002/sscp.202200134.
Podar AS, Semeniuc CA, Ionescu SR, Socaciu M-I, Fogarasi M, Fărcaș AC, Vodnar DC, Socaci SA. An Overview of Analytical Methods for Quantitative Determination of Coenzyme Q10 in Foods. Metabolites, 13, 272 (2023) https://doi.org/10.3390/metabo13020272.
Nakov N, Acevska J, Brezovska K, Kavrakovski Z, Dimitrovska A. Green Strategies toward Eco-Friendly HPLC Methods in Pharma Analysis. High Performance Liquid Chromatography - Recent Advances and Applications. In tech Open, (2023), https://doi.org/10.5772/intechopen.110035.
Ameen SA, Pappula N. Analytical QBD Approach to Redefine the Quality of Pharmaceuticals: A Review. Journal of Pharmaceutical Research, 22, 178–85 (2023) https://doi.org/10.18579/jopcr/v22.4.81.
Xuan DT, Nguyen HMT, Hoang VD. Recent applications of analytical quality-by-design methodology for chromatographic analysis: A review. Chemometrics and Intelligent Laboratory Systems, 254, 105243 (2024) https://doi.org/10.1016/j.chemolab.2024.105243.
Bairagi A, Kothrukar R, Chikhale H, Kosanam S, Borse L. AQbD-novel strategy for analytical methods. Futur J Pharm Sci, 10, 138 (2024) https://doi.org/10.1186/s43094-024-00706-1.
Nunsavathu SN, Rajaganapathy K. A Review on QbD Approach in Analytical Method Development and Validation . International Journal of Pharmaceutical Quality Assurance, 15, 1707–13 (2024) https://doi.org/10.25258/ijpqa.15.3.93.
Giri S, Varshney KK, Srivastava R. Therapeutic Potential and Prospects of L-arginine in Various Diseases and its Clinical Intervention. Curr Drug ther, 19, 529–45 (2024) https://doi.org/10.2174/0115748855260802231019072509.
Matta A, Sundararajan R. Green Analytical Stability Indicating UHPLC Method for the Quantification of Related Impurities in Vonoprazan Formulation Applying Analytical Quality by Design. Sep Sci Plus, 8, (2025) https://doi.org/10.1002/sscp.70032.
Goraksh TA, Gitaram VD. QbD-Based RP-HPLC Method Development and Validation for Quantitation of Rimegepant in Standard and Pharmaceutical Formulations. International Journal of Pharmaceutical Research and Applications, 10, 942–5 (2025) https://doi.org/10.35629/4494-1002942945.
Raju C, Bojan S, Chinthaginjala C, Dravidamani M, Dommaraju AK, Raghunathan K. Analytical Quality by Design Assisted Optimization of RP-HPLC Method for the Estimation of Palovarotene Drug Substance and Drug Product by Box–Behnken Design. Biosci Biotechnol Res Asia, 22, 313–24 (2025) https://doi.org/10.13005/bbra/3363.
Kudalkar S, Arya S, Pradhan M. Coenzyme Q10: A Comprehensive Review of Its Roles in Mitochondrial Health and Systemic Function. Int J Health Sci Res, 15, 106–15 (2025) https://doi.org/10.52403/ijhsr.20250914.
Valvi AM, Shelke RU, Ghodke SS, Rishipathak DD. Quality By Design and Green Analytical Chemistry: A Review of Novel Approaches to Chromatographic Method Development. Biosci Biotechnol Res Asia, 22, 497–520 (2025) https://doi.org/10.13005/bbra/3379.
Tamboli FA, Mane PP, Babaleshwar RB, Mane DM, Attar PS, Disale PP, Patil PA, Parkhi SM, Rathod MP. From concept to compliance: The role of QbD in modern product development. Current Trends in Pharmacy and Pharmaceutical Chemistry, 6, 168–75 (2025) https://doi.org/10.18231/j.ctppc.2024.032.
Published
How to Cite
Issue
Section
Copyright (c) 2025 Vishal Kumar Pathak, Phool Chandra, Bhupendra Singh

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
















