QbD-guided HPTLC method development and validation for quantitative estimation of anticancer drugs
DOI:
https://doi.org/10.69857/joapr.v13i5.1465Keywords:
HPTLC, Anticancer drug, Method Validation, Quality by Design, Factorial DesignAbstract
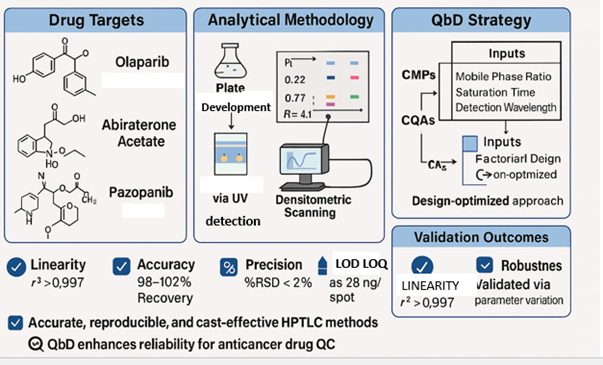
Background: Olaparib, Abiraterone acetate, and Pazopanib are critical anticancer agents used in the treatment of breast/ovarian, prostate, and renal cancers, respectively. Ensuring their quality through precise, cost-effective analytical techniques is vital for routine quality control (QC). Given their clinical importance, a robust method capable of quantifying these drugs individually and indicating their stability under stress is highly desirable. Methodology: High-performance thin-layer chromatography (HPTLC) methods were developed for each drug, guided by a Quality by Design (QbD) approach. A 2³ factorial design was employed to optimize three critical method parameters: mobile phase composition, chamber saturation time, and detection wavelength. Chromatographic analysis was performed on aluminum-backed silica gel plates with detection wavelengths set at 278 nm (Olaparib), 255 nm (Abiraterone acetate), and 254 nm (Pazopanib). Method validation followed ICH Q2(R2) guidelines, assessing linearity, accuracy, precision, LOD/LOQ, specificity, and robustness. Forced degradation under acidic, basic, oxidative, thermal, and photolytic conditions was evaluated to assess the stability-indicating capability. Results and Discussion: All methods exhibited strong linearity (r² > 0.997), high accuracy (98–102% recovery), and precision (%RSD < 2). Sensitivity was excellent, with LOD as low as 28 ng/spot for Olaparib. Under stress, degradation ranged from 5% to 20%, with distinct degradant peaks and peak purity indices of> 0.995, confirming no co-elution and indicating stability. The validated methods were successfully applied to stressed marketed formulations. Conclusion: The QbD-optimized HPTLC methods are accurate, economical, and stability-indicating, making them suitable for routine QC of Olaparib, Abiraterone acetate, and Pazopanib in pharmaceutical dosage forms.
Downloads
References
Zhang W, He L, Shi Y, Gong J, Liu Y, Shao L. Development and validation of an HPTLC method for simultaneous estimation of multiple drugs in pharmaceutical formulations. J Chromatogr Sci, 58(2), 214–221 (2020) https://doi.org/10.1093/chromsci/bmz080
Pramod K, Tahir MA, Charoo NA, Ansari SH, Ali J. Pharmaceutical product development: a quality by design approach. Int J Pharm Investig, 6(3), 129–138 (2016) https://doi.org/10.4103/2230-973X.187350
Jethava P, Patel N. Analytical Quality by Design (AQbD): a comprehensive review with emphasis on risk assessment approaches. Int J Res Pharm Allied Sci, 4(8), 1–16 (2025) https://doi.org/10.71431/IJRPAS.2025.4801
Sahu AK, Rao KS, Panda S. Stability-indicating HPTLC method for the estimation of Olaparib. Indian J Pharm Sci, 82(6), 1061–1068 (2020) https://doi.org/10.36468/pharmaceutical-sciences.663
Kamble MB, Chaudhari SP, Pawar AY, Thakare VM. Stability-indicating RP-HPLC method for pazopanib hydrochloride. J Appl Pharm Sci, 11(3), 134–140 (2021) https://doi.org/10.7324/JAPS.2021.110316
Sharma T, Khurana RK, Borges B, Kaur R, Katare OP, Singh B. An HPTLC densitometric method for simultaneous quantification of Sorafenib Tosylate and Chrysin: analytical method development, validation and applications. Microchem J, 162, 105821 (2021) https://doi.org/10.1016/j.microc.2020.105821
Deshmukh VS, Wankhede SB. QbD-based HPTLC method development for estimation of drugs in bulk and dosage form. J Appl Pharm Sci, 13(2), 155–162 (2023) https://doi.org/10.7324/JAPS.2023.130217
Malik Z, Parveen R, Zahiruddin S, Gautam G, Husain SA, Ahmad S. HPTLC stability-indicating analytical method of Andrographolide and 5-fluorouracil with network pharmacology analysis against cancer. Comb Chem High Throughput Screen, 27(6), 894–909 (2024) https://doi.org/10.2174/1386207327666230202120747
Parmar VK, Mistry D, Patel MD, Saradhara D. DoE-based development and validation of HPTLC method for simultaneous estimation of Curcumin and Naringin in topical gel formulation. Anal Chem Lett, 14(1), 48–68 (2024) https://doi.org/10.1080/22297928.2024.2314527
Lee SH, Kim JK, Jee JP. Quality by Design (QbD) application for the pharmaceutical development process. J Pharm Investig, 52, 649–682 (2022) https://doi.org/10.1007/s40005-022-00575-x
Bang PP, Bhatt HG. Development of green RP- and green NP-HPTLC methods for estimation of Lenvatinib and comparative evaluation by AGREE. ACS Sustain Chem Eng, 11(6), 2249–2263 (2023) https://doi.org/10.1021/acssuschemeng.2c06163
P N S, Adikay S. A QbD-based stability-indicating RP-HPLC method for Larotrectinib: degradation kinetics and integrated white, green, and blue analytical assessment. J Appl Pharm Res, 13(4), 143–161 (2025) https://doi.org/10.69857/joapr.v13i4.1436
Joshi P, Panchal H. HPTLC and AQbD: a new fusion in analytical research. Res J Pharm Technol, 15(6), 2561–2567 (2022) https://doi.org/10.52711/0974-360X.2022.00429
Chaudhary MK, Misra A, Srivastava S. A multi-analyte HPTLC estimation of marker compounds in Pueraria tuberosa (Willd.) DC.: application of AQbD approach for method optimization and greenness assessment. Microchem J, 199, 109988 (2024) https://doi.org/10.1016/j.microc.2024.109988
Koradia SK, Patel M, Sen AK, Sen DB, Pradhan P. Analytical quality by design-based thin-layer chromatography method development and validation for assay and content uniformity testing of the anti-neoplastic drug Axitinib in tablet formulation. Sep Sci Plus, 7(3), 2300176 (2024) https://doi.org/10.1002/sscp.202300176
Chiarentin L, Gonçalves C, Augusto C, Miranda M, Cardoso C, Vitorino C. Drilling into “Quality by Design” approach for analytical methods. Crit Rev Anal Chem, 54(8), 3478–3519 (2024) https://doi.org/10.1080/10408347.2023.2253321
Canil M, De Nicola M, Cattaneo D, Clementi E, Falvella FS. LC–MS/MS method for the quantification of PARP inhibitors Olaparib, Rucaparib and Niraparib in human plasma and dried blood spot: development, validation and clinical validation for therapeutic drug monitoring. Pharmaceutics, 15(5), 1524 (2023) https://doi.org/10.3390/pharmaceutics15051524
Gandhi J, Pandya R. RP-HPLC method development and validation for Olaparib in tablet dosage form. Res J Pharm Technol, 14(10), 3573–3576 (2021) https://doi.org/10.52711/0974-360X.2021.00623
Mhaske DV, Dhaneshwar SR. Stability-indicating RP-HPLC method for estimation of Abiraterone acetate. J Pharm Anal, 10(1), 69–76 (2020) https://doi.org/10.1016/j.jpha.2020.04.001
Gowda DV, Shree JA, Rakesh S, Ahmed MG. UV spectrophotometric method for estimation of Abiraterone acetate in bulk and tablet dosage form. Int J ChemTech Res, 13(1), 172–176 (2020) https://doi.org/10.20902/IJCTR.2019.130125
Beg S, Malik AK, Afzal O, Altamimi AS, Kazmi I, Al-Abbasi FA, Almalki WH, Barkat MA, Kawish SM, Pradhan DP, Rahman M. Systematic development and validation of a RP-HPLC method for estimation of Abiraterone acetate and its degradation products. J Chromatogr Sci, 59(1), 79–87 (2021) https://doi.org/10.1093/chromsci/bmaa080
Bagada H, Vanzara R, Palva R, Karkhanis V. Application of Quality by Design in the development of HPTLC method for the estimation of Ceritinib in bulk and synthetic mixture. Discov Chem, 1, 46 (2024) https://doi.org/10.1007/s44371-024-00036-4
Alam P, Salem-Bekhit MM, Al-Joufi FA, Alqarni MH, Shakeel F. Quantitative analysis of Cabozantinib in pharmaceutical dosage forms using green RP-HPTLC and green NP-HPTLC methods: a comparative evaluation. Sustain Chem Pharm, 21, 100413 (2021) https://doi.org/10.1016/j.scp.2021.100413
Yang S, Hu X, Zhu J, Zheng B, Bi W, Wang X, Wu J, Mi Z, Wu Y. Aspects and implementation of pharmaceutical Quality by Design from conceptual frameworks to industrial applications. Pharmaceutics, 17(5), 623 (2025) https://doi.org/10.3390/pharmaceutics17050623
Published
How to Cite
Issue
Section
Copyright (c) 2025 Bhavik Jani, Hitesh Vekariya

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
















