Optimization of fast melting olanzapine tablets using solid dispersion and response surface methodology
DOI:
https://doi.org/10.69857/joapr.v13i5.1191Keywords:
Olanzapine, phase solubility, sodium starch glycolate, fast meltingAbstract
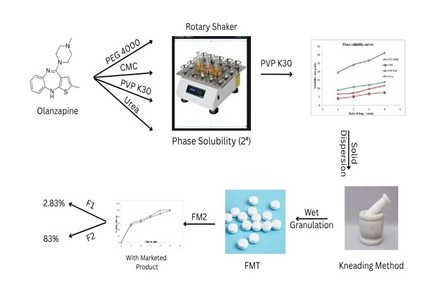
Background: Olanzapine is a poorly water-soluble, anti-psychotic drug that belongs to the class of thiobenzodiazepines, which has a bioavailability of 60 – 65%. The purpose of this research work is to enhance the solubility of olanzapine by the solid dispersion technique using different water-soluble carriers, using a phase solubility study using a 24 factorial design, and to incorporate the solid dispersion of olanzapine to formulate fast-melting tablets using different superdisintegrants. Methodology: The fast-melting tablets were prepared using the wet granulation technique and optimized through a 23 full factorial design. The independent variables are sodium starch glycolate (X1), sodium carboxymethyl cellulose (X2), and the method of preparation of the solid dispersion (X3). The dependent variables are hardness (Y1), friability (Y2), disintegration time (Y3), and in vitro drug release studies (Y4). The kinship between independent and dependent variables was demonstrated using contour diagrams. Additionally, the prepared fast-melting tablets were analysed for their weight variation, drug content uniformity, and other dependent variables. Results and Discussion: The fast-melting tablets (batch F6) were considered desirable based on their drug content (99.5% & drug release of 99.3% in 20 minutes, following first–order and Higuchi kinetics. The difference factor f1and f2 similarity factor were found to be 2.43% and 83%, respectively, for the optimized formulation FM2, and the drug release was greater than that of the marketed product. Conclusion: It is evident that the optimized formulation FM2 appears to be a promising system that facilitates the rapid release of olanzapine compared to other formulations.
Downloads
References
Kolli P, Kelley G, Rosales M, Faden J, Serdenes R. Olanzapine pharmacokinetics: A clinical review of current insights and remaining questions. Pharmacogenomics Pers. Med., 16, 1097-1108 (2023) https://doi.org/10.2147/pgpm.s391401
Bhujbala SV, Mitra B, Jain U, Gong Y, Agrawal A, Karki S, et al. Pharmaceutical amorphous solid dispersion: A review of manufacturing strategies. Acta Pharm. Sin. B, 11(8), 2505-2536 (2021) https://doi.org/10.1016/j.apsb.2021.05.014
Gnanaprakash M, Gomathi M, Jothimanivannan C, Vignesh M, Thinakaran P, Dinesh Kumar L. Fast dissolving tablet – A review. Int. J. Pharm. Sci. Rev. Res., 79(1), 7-14 (2023) http://dx.doi.org/10.47583/ijpsrr.2023.v79i01.002
Mahesh M, Harish KH, Dasankoppa FS, Kshatriya K, Latwade R, Patil P, et al. Formulation and evaluation of fast disintegrating tablets of antihistamine drug using natural super disintegrants. J. Young Pharm., 16(4), 762-771 (2024) https://doi.org/10.5530/jyp.2024.16.96
Bellaiah PG, Hagalavadi NS, Madalli RK. 23 factorial design: An approach for formulation of solid lipid nanoparticles of etravirine for oral administration. Indian J Pharm Sci., 84(3), 712-722 (2022) https://doi.org/10.36468/pharmaceutical-sciences.966
Wagh VT, Gilhotra RM, Rajendra D, et al. Solid dispersion technique: A platform for enhancement dissolution rate of valsartan poorly water soluble drug. Int. J. Pharm. Qual. Assur., 11, 21-24 (2020) https://doi.org/10.25258/ijpqa.11.1.3
Supare V, Wadher K, Umekar M. Experimental design: Approaches and applications in development of pharmaceutical drug delivery system. J Drug Deliv Ther., 11, 154-161 (2021) https://doi.org/10.22270/jddt.v11i4-S.4908
Eisa AM, et al. Formulation and evaluation of fast dissolving tablets of haloperidol solid dispersion. Saudi Pharm. J., 30, 1589-1602 (2022) https://doi.org/10.1016/j.jsps.2022.09.002
Raj GP, Bhavya G, Vardhana MGH, Gowda HUJ. A review of formulation techniques and evaluation parameters of fast dissolving tablets. J. Pharma Insights Res., 2(4), 212-217 (2024) https://doi.org/10.69613/vx91cb41
Vadaga AK, Gudla SS, Nareboina GSK, Gubbala H, Golla B. Comprehesive review on modern techniques of granulation in pharmaceutical solid dosage forms. Intelligent Pharmacy, 2(5), 609-629 (2024) https://doi.org/10.1016/j.ipha.2024.05.006
Aleksic I, Glisic T, Cirin-Varadan S, Djuris M, Djuris J, Parojcic J. Evaluation of the potential of novel co-processed excipients to enable direct compression and modified release of ibuprofen. Pharmaceutics, 16(11), 1473 (2024) https://doi.org/10.3390/pharmaceutics16111473
Zheng Y, Yang X, Valdes M, Davis AM, Heck PR. Volume measurement of microparticles using SEM photogrammetry and 3-D reconstruction. Meteorit. Planet. Sci., 60(5), 1227-1235 (2025) https://doi.org/10.1111/maps.14351
Maheshwari S, Singh A, Varshney AP, Sharma A. Advancing oral drug delivery: The science of fast dissolving tablets. Intelligent Pharmacy, 2, 580-587 (2024) https://doi.org/10.1016/j.ipha.2024.01.011
Rakhi R, Singh S, Kumar A, Mishra AK. Formulation and evaluation of fast-dissolving tablets of febuxostat using co-processed excipients. J. Young Pharm., 12(3), 231-237 (2020) http://dx.doi.org/10.2174/1872211314666191224121044
Mahesh PG, Raman SG. Formulation and evaluation of fast dissolving tablet of clopidogrel. Res. J. Pharm. Technol., 13(9), 4084-4086 (2020) https://doi.org/10.5958/0974-360X.2020.00721.0
Kaity S, Sah SK, Karanwad T, Banerjee S. Bootstrap statistics and its application in disintegration and dissolution data analysis. Mol. Pharm., 20(8), 3791-3803 (2023) https://doi.org/10.1021/acs.molpharmaceut.3c00222
Published
How to Cite
Issue
Section
Copyright (c) 2025 Revathi Sundaramoorthi, Anitha Pavadai, Jenifer Sathappan, Senthil Rajan Sivakumar

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
















