Design and optimization of chitosan microspheres loaded with green tea phytosomes for sustained release
DOI:
https://doi.org/10.69857/joapr.v13i5.1188Keywords:
Microsphere, phytosome, green tea, polyphenols, in vitro washed offAbstract
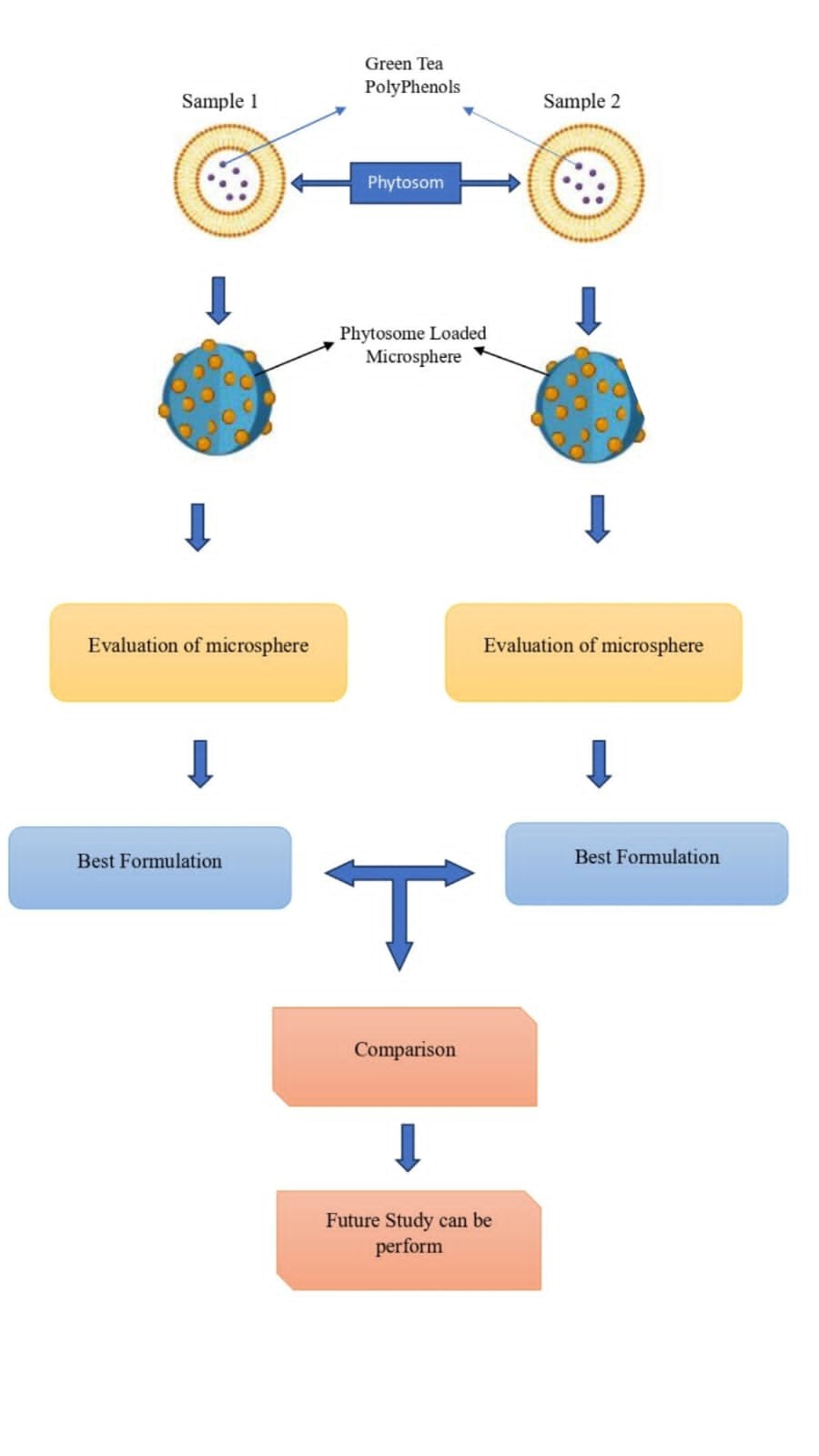
Background: Phytosomes are nanovesicular systems that integrate plant extracts with phospholipids to improve the solubility, stability, and bioavailability of phytoconstituents. Green tea (Camellia sinensis) is rich in polyphenols such as epigallocatechin gallate (EGCG) and epigallocatechin (EGC), which possess significant therapeutic potential but are limited by poor absorption and stability. The present study aimed to formulate and evaluate green tea extract–loaded phytosome-incorporated microspheres with desirable physicochemical characteristics for sustained delivery. Methodology: Phytosomes were prepared using the thin-layer hydration method with varying molar ratios (0.5–1.0) of phospholipids to standardized green tea extract (sample 1 and sample 2). The optimized phytosomes were further encapsulated into microspheres via emulsion cross-linking, employing different concentrations of glutaraldehyde and polymer to obtain nine formulations. Design Expert software was applied for optimization, and the microspheres were evaluated for micrometric properties, entrapment efficiency, drug loading, drug release, swelling behaviour, mucoadhesion, stability, and surface morphology. Results and Discussion: The prepared microspheres exhibited a spherical morphology with satisfactory physicochemical properties. Among the formulations, batch F3 of sample 1 demonstrated the most promising results, achieving 87% yield, 77% drug entrapment efficiency, 30% drug loading, and 91.87% cumulative drug release up to 9 hours, along with favorable swelling and mucoadhesion properties. Stability studies further confirmed the reliability of the formulation. Conclusion: Overall, the developed phytosome-loaded microspheres of green tea extract exhibited an improved release profile, stability, and potential fwr sustained drug delivery, suggesting their applicability in enhancing the therapeutic efficacy of green tea polyphenols.
Downloads
References
Fayed MH, Alalaiwe A, Almalki ZS, Helal DA. Design Space Approach for the Optimization of Green Fluidized Bed Granulation Process in the Granulation of a Poorly Water-Soluble Fenofibrate Using Design of Experiment. Pharmaceutics, 14, 1471 (2022) https://doi.org/10.3390/pharmaceutics14071471.
Sharma A, Arora K, Mohapatra H, et al. Supersaturation-Based Drug Delivery Systems: Strategy for Bioavailability Enhancement of Poorly Water-Soluble Drugs. Molecules 27, 2969 (2022) https://doi.org/10.3390/molecules27092969
Lazar AR, Puscas A, Tanislav AE, Muresan V. Bioactive compounds delivery and bioavailability in structured edible oils systems. Compr. Rev. food Sci. food Saf., 23, 1-45 (2024) https://doi.org/10.1111/1541-4337.70020.
Koppula S, Shaik B, Maddi S. Phytosomes as a New Frontier and Emerging Nanotechnology Platform for Phytopharmaceuticals: Therapeutic and Clinical Applications. Phytother. Res., 39, 2217-2249 (2025) https://doi.org/10.1002/ptr.8465.
Al-Nemrawi NK, Abu Dayah A, Darweesh R. Poly(Lactic-co-Glycolic Acid Nanoparticles Loaded with Docetaxel and Coated with Chitosan, Carboxymethyl Chitosan, or Glycol Chitosan. Curr. Pharm. Biotechnol., 2590 - 2603 (2024) https://doi.org/10.2174/0113892010335722240923110808.
Liu X, Obacz J, Emanuelli G, Chambers JE, Abreu S, Chen X et al. Enhancing Drug Delivery Efficacy Through Bilayer Coating of Zirconium-Based Metal-Organic Frameworks: Sustained Release and Improved Chemical Stability and Cellular Uptake for Cancer Therapy. Chem. Mater., 36, 3588–3603 (2024) https://doi.org/10.1021/acs.chemmater.3c02954.
Chen W, Li H, Zhang X, Sang Y, Nie Z. Microfluidic preparation of monodisperse PLGA-PEG/PLGA microspheres with controllable morphology for drug release. Lab Chip, 24, 4623-4631 (2024) https://doi.org/10.1039/d4lc00486h.
da Silva AF, Moreira AF, Miguel SP, Coutinho P. Recent advances in microalgae encapsulation techniques for biomedical applications. Adv. Colloid Interface Sci., 333, 1-9 (2024) https://doi.org/10.1016/j.cis.2024.103297.
Pan CT, Chien ST, Chiang TC, Yen CK, Wang SY, Wen ZH, Yu CY, Shiue YL. Optimization of the spherical integrity for sustained-release alginate microcarriers-encapsulated doxorubicin by the Taguchi method. Sci. Rep., 10, 21758 (2020) https://doi.org/10.1038/s41598-020-78813-1.
Pandey V, Rathee S, Sen D, Jain SK, Patil UK. Phytovesicular Nanoconstructs for Advanced Delivery of Medicinal Metabolites: An In-Depth Review. Curr. Drug Targets, 25, 847–65 (2024) https://doi.org/10.2174/0113894501310832240815071618.
Park H. Exploring the Effects of Process Parameters during W/O/W Emulsion Preparation and Supercritical Fluid Extraction on the Protein Encapsulation and Release Properties of PLGA Microspheres. Pharmaceutics, 16, 302 (2024) https://doi.org/10.3390/pharmaceutics16030302.
Cholidis P, Kranas D, Chira A, Galouni EA, Adamantidi T, Anastasiadou C, Tsoupras A. Shrimp Lipid Bioactives with Anti-Inflammatory, Antithrombotic, and Antioxidant Health-Promoting Properties for Cardio-Protection. Mar. Drugs, 22, 554 (2024) https://doi.org/10.3390/md22120554.
Chen Y, Tang Y, Li Y, Rui Y, Zhang P. Enhancing the Efficacy of Active Pharmaceutical Ingredients in Medicinal Plants through Nanoformulations: A Promising Field. Nanomater. (Basel, Switzerland), 14, 1598 (2024) https://doi.org/10.3390/nano14191598.
Permana AD, Utami RN, Courtenay AJ, Manggau MA, Donnelly RF, Rahman L. Phytosomal nanocarriers as platforms for improved delivery of natural antioxidant and photoprotective compounds in propolis: An approach for enhanced both dissolution behaviour in biorelevant media and skin retention profiles. J. Photochem. Photobiol. B., 205, 111846 (2020) https://doi.org/10.1016/j.jphotobiol.2020.
Kothawade S, Bhange M, Pande VV. Advancements in Phytosomal Therapies for Liver Cancer: A Comprehensive Review. Curr. Cancer Drug Targets, 25, 1066 - 1082 (2024) https://doi.org/10.2174/0115680096319007240703072650.
Rana L, Harwansh RK, Deshmukh R. Recent Updates on Phytopharmaceuticals-Based Novel Phytosomal Systems and Their Clinical Trial Status: A Translational Perspective. Crit. Rev. Ther. Drug Carrier Syst., 42, 1–54 (2025) https://doi.org/10.1615/CritRevTherDrugCarrierSyst.v42.i1.10.
Yuan Z, Wan Z, Wei P, Lu X, Mao J, Cai Q, Zhang X, Yang X. Dual-Controlled Release of Icariin/Mg(2+) from Biodegradable Microspheres and Their Synergistic Upregulation Effect on Bone Regeneration. Adv. Healthc. Mater., 9, e2000211 (2020) https://doi.org/10.1002/adhm.202000211.
Zhang Q, Du Y, Yu M, Ren L, Guo Y, Li Q, Yin M, Li X, Chen F. Controlled release of dinotefuran with temperature/pH-responsive chitosan-gelatin microspheres to reduce leaching risk during application. Carbohydr. Polym., 277, 118880 (2022) https://doi.org/10.1016/j.carbpol.2021.118880.
Sole-Enrech G, Cano-Corres R, Aparicio-Calvente MI, Spataro N. Elimination of lipaemic interference by high-speed centrifugation. Biochem. medica, 33, 10703 (2023) https://doi.org/10.11613/BM.2023.010703.
Ashikhmin A, Piskunov M, Kochkin D, Ronshin F, Chen L. Droplet Microfluidic Method for Estimating the Dynamic Interfacial Tension of Ion-Crosslinked Sodium Alginate Microspheres. Langmuir, 15906–15917 (2024) https://doi.org/10.1021/acs.langmuir.4c01940.
Zhou J, Walker J, Ackermann R, Olsen K, Hong JKY, Wang Y, Schwendeman SP. Effect of Manufacturing Variables and Raw Materials on the Composition-Equivalent PLGA Microspheres for 1-Month Controlled Release of Leuprolide. Mol. Pharm., 17, 1502–15 (2020) https://doi.org/10.1021/acs.molpharmaceut.9b01188.
Gelli R, Mugnaini G, Bolognesi T, Bonini M. Cross-linked Porous Gelatin Microparticles with Tunable Shape, Size, and Porosity. Langmuir, 37, 12781–9 (2021) https://doi.org/10.1021/acs.langmuir.1c01508.
Ramkanth S, Anitha P, Gayathri R, Mohan S, Babu D. Formulation and design optimization of nano-transferosomes using pioglitazone and eprosartan mesylate for concomitant therapy against diabetes and hypertension. Eur. J. Pharm. Sci. Off. J. Eur. Fed. Pharm. Sci., 162, 105811 (2021) https://doi.org/10.1016/j.ejps.2021.105811.
Tafere C, Yilma Z, Abrha S, Yehualaw A. Formulation, in vitro characterization and optimization of taste-masked orally disintegrating co-trimoxazole tablet by direct compression. PLoS One, 16, e0246648 (2021) https://doi.org/10.1371/journal.pone.0246648.
Zeng H-L, Qiu Q, Fu T-X, Deng A-P, Xie X-Y. Development and optimization of sustained release triptolide microspheres. PLoS One, 18, e0292861 (2023) https://doi.org/10.1371/journal.pone.0292861.
Verberk IM, Nossent EJ, Bontkes HJ, Teunissen CE. Pre-analytical sample handling effects on blood cytokine levels: quality control of a COVID-19 biobank. Biomark. Med., 15, 987–97 (2021) https://doi.org/10.2217/bmm-2020-0770.
Khan S, Arshad S, Arif A, Tanveer R, Amin ZS, Abbas S, Maqsood A, Raza M, Munir A, Latif A, Habiba M, Afzal M. Trypsin Inhibitor Isolated From Glycine max (Soya Bean) Extraction, Purification, and Characterization. Dose. Response., 20, 15593258221131462 (2022) https://doi.org/10.1177/15593258221131462.
Maclean N, Armstrong JA, Carroll MA, Salehian M, Mann J, Reynolds G, Johnston B, Markl D. Flexible modelling of the dissolution performance of directly compressed tablets. Int. J. Pharm., 656, 124084 (2024) https://doi.org/10.1016/j.ijpharm.2024.124084.
Chamsai B, Opanasopit P, Samprasit W. Fast disintegrating dosage forms of mucoadhesive-based nanoparticles for oral insulin delivery: Optimization to in vivo evaluation. Int. J. Pharm., 647, 123513 (2023) https://doi.org/10.1016/j.ijpharm.2023.123513.
González-González O, Ramirez IO, Ramirez BI, O’Connell P, Ballesteros MP, Torrado JJ, Serrano DR. Drug Stability: ICH versus Accelerated Predictive Stability Studies. Pharmaceutics, 14, 2324 (2022) https://doi.org/10.3390/pharmaceutics14112324.
Ullah F, Shah KU, Shah SU, Nawaz A, Nawaz T, Khan KA, Alserihi RF, Tayeb HH, Tabrez S, Alfatama M. Synthesis, Characterization and In Vitro Evaluation of Chitosan Nanoparticles Physically Admixed with Lactose Microspheres for Pulmonary Delivery of Montelukast. Polymers (Basel)., 14, 3564 (2022) https://doi.org/10.3390/polym14173564.
Li Y, Keqi W, Wang G. Evaluating disease similarity based on gene network reconstruction and representation. Bioinformatics, 37, 3579–87 (2021) https://doi.org/10.1093/bioinformatics/btab252.
Akhlaq M, Azad AK, Ullah I, Nawaz A, Safdar M, Bhattacharya T, Uddin ABMH, Abbas SA, Mathews A, Kundu SK, Miret MM, Murthy HCA, Nagaswarupa HP. Methotrexate-Loaded Gelatin and Polyvinyl Alcohol (Gel/PVA) Hydrogel as a pH-Sensitive Matrix. Polymers (Basel)., 13, 2300 (2021) https://doi.org/10.3390/polym13142300.
Kulkarni R, Fanse S, Burgess DJ. Mucoadhesive drug delivery systems: a promising non-invasive approach to bioavailability enhancement. Part I: biophysical considerations. Expert Opin. Drug Deliv., 20, 395–412 (2023) https://doi.org/10.1080/17425247.2023.2181331.
Zhang Q, Li X, Jasti BR. Role of physicochemical properties of some grades of hydroxypropyl methylcellulose on in vitro mucoadhesion. Int. J. Pharm., 609, 121218 (2021) https://doi.org/10.1016/j.ijpharm.2021.121218.
Kiss T, Ambrus R, Abdelghafour MM, Zeiringer S, Selmani A, Roblegg E, Budai-Szűcs M, Janovák L, Lőrinczi B, Deák Á, Bernkop-Schnürch A, Katona G. Preparation and detailed characterization of the thiomer chitosan-cysteine as a suitable mucoadhesive excipient for nasal powders. Int. J. Pharm., 626, 122188 (2022) https://doi.org/10.1016/j.ijpharm.2022.122188.
Jabar A, Madni A, Bashir S, Tahir N, Usman F, Rahim MA, Jan N, Shah H, Khan A, Khan S. Statistically optimized pentazocine loaded microsphere for the sustained delivery application: Formulation and characterization. PLoS One, 16, e0250876 (2021) https://doi.org/10.1371/journal.pone.0250876.
Perveen R, Latif F, Abbas M, Ayub K, Sarfaraz S, Saeed M, Rana S, Al-Rashida M, Nawaz MA, Hameed A. Optimization study and application of box-behnken model for probing eggshell supported transition metals based catalysts to synthesize hydrazone & dihydropyrimidinones. Sci. Rep., 14, 23270 (2024) https://doi.org/10.1038/s41598-024-74876-6.
Attia MS, Radwan MF, Ibrahim TS, Ibrahim TM. Development of Carvedilol-Loaded Albumin-Based Nanoparticles with Factorial Design to Optimize In Vitro and In Vivo Performance. Pharmaceutics, 15, 1425 (2023) https://doi.org/10.3390/pharmaceutics150514
Miu D-M, Pavaloiu RD, Sha’at F, Vladu M-G, Neagu G, Manoiu V-S, Eremia M-C. Preparation and Optimization of a Polyhydroxyoctanoate-Hydroxyapatite Composite Available to Scaffolds in Implantable Devices. Molecules, 30, 730 (2025) https://doi.org/10.3390/molecules30030730.
Frenț OD, Duteanu N, Teusdea AC, Ciocan S, Vicaș L, Jurca T, Muresan M, Pallag A, Ianasi P, Marian E. Preparation and Characterization of Chitosan-Alginate Microspheres Loaded with Quercetin. Polymers, 14, 490 (2022) https://doi.org/10.3390/polym14030490.
Martinez-Ballesta, M, Gil-Izquierdo A, Garcia-Viguera C, Dominguez-Perles R. Nanoparticles and Controlled Delivery for Bioactive Compounds: Outlining Challenges for New “Smart-Foods” for Health. Foods, 7, 72 (2018) https://doi.org/10.3390/foods7050072.
Barani M, Sangiovanni E, Angarano M, Rajizadeh MA, Mehrabani M, Piazza S, Gangadharappa HV, Pardakhty A, Mehrbani M, Dell'Agli M, Nematollahi MH. Phytosomes as Innovative Delivery Systems for Phytochemicals: A Comprehensive Review of Literature. Int J Nanomedicine, 16, 6983-7022 (2021) https://doi.org/10.2147/IJN.S318416.
Guo Y, Luo J, Tan S, Otieno BO, Zhang Z. The applications of Vitamin E TPGS in drug delivery. Eur J Pharm Sci. 13, 49 (2013):175-86 https://doi.org/10.1016/j.ejps.2013.02.006.
P K, R K. Phytosome Technology: A Novel Breakthrough for the Health Challenges. Cureus. 30, 16 (2024) https://doi.org/10.7759/cureus.68180.
Published
How to Cite
Issue
Section
Copyright (c) 2025 Juti Rani Devi, Trishna Das, Bhupen Kalita, Bhargab Jyoti Sahariah

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
















